 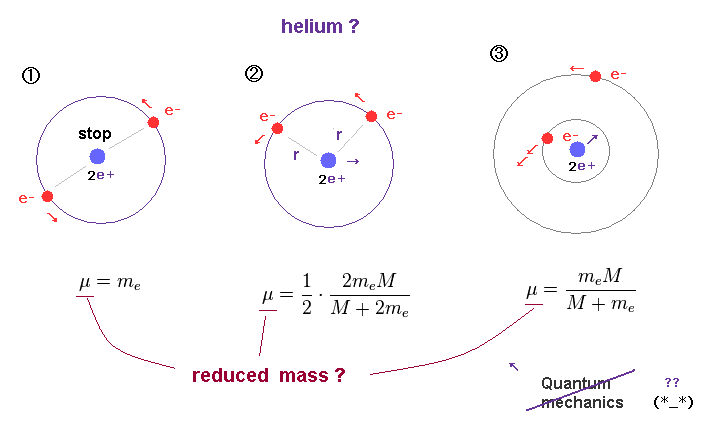
The first step of the proton–proton chain reaction is a two-stage process first, two protons fuse to form a diproton:įollowed by the immediate beta-plus decay of the diproton to deuterium: Is an intermediate in the first step of the proton–proton chain reaction. A similar reaction has also been observed from 8 Nucleus, which quickly decayed into two protons. The two remaining protons could be simultaneously ejected from the target as a 2 Nucleus can donate all four of its neutrons to the hydrogen. Nuclei were directed at a cryogenic hydrogen target to produce 5 The new experiment showed that the two protons were initially ejected together, correlated in a quasibound 1S configuration, before decaying into separate protons much less than a nanosecond later.įurther evidence comes from RIKEN in Japan and the Joint Institute for Nuclear Research in Dubna, Russia, where beams of 6 Nucleus, plus two protons detected exiting from the same direction. As in the earlier experiment at Oak Ridge, the 18 Nucleus into a highly unstable condition. The second collision had the effect of exciting the 18 These nuclei then collided with a foil of lead. This collision converted some of the heavier neon nuclei in the beam into 18 Ions was directed at a target of beryllium foil. Was found in 2008 at the Istituto Nazionale di Fisica Nucleare, in Italy. The experiment was not sensitive enough to establish which of these two processes was taking place. Alternatively, the protons may be emitted separately but simultaneously-so-called "democratic decay". Nucleus-which then decays into separate protons. The neon nucleus might eject a "diproton"-a pair of protons bundled together as a 2 There are two ways in which the two-proton emission may proceed. Any protons ejected from the target itself were identified by their characteristic energies. , which then decayed into oxygen and two protons. The team fired a beam of fluorine ions at a proton-rich target to produce 18 This means that the two protons are ejected simultaneously. Galindo-Uribarri and co-workers chose an isotope of neon with an energy structure that prevents it from emitting protons one at a time. The team led by Alfredo Galindo-Uribarri of the Oak Ridge National Laboratory announced that the discovery will help scientists understand the strong nuclear force and provide fresh insights into the creation of elements inside stars. In 2000, physicists first observed a new type of radioactive decay in which a nucleus emits two protons at once-perhaps a 2 Its instability is due to spin–spin interactions in the nuclear force, and the Pauli exclusion principle, which forces the two protons to have anti-aligned spins and gives the diproton a negative binding energy. According to theoretical calculations, it would have been much more stable (although still undergoing β + decay to deuterium) if the strong interaction had been 2% greater. Its nucleus, a diproton, consists of two protons with no neutrons. 
Is an extremely unstable isotope of helium. ^ a b Produced during Big Bang nucleosynthesisĪre the only stable nuclides with more protons than neutrons.^ Intermediate in the proton–proton chain reaction.^ # – Values marked # are not purely derived from experimental data, but at least partly from trends of neighboring nuclides (TNN).^ ( ) spin value – Indicates spin with weak assignment arguments.^ Bold symbol as daughter – Daughter product is stable.^ ( ) – Uncertainty (1 σ) is given in concise form in parentheses after the corresponding last digits.Dilution refrigerators take advantage of the immiscibility of these two isotopes to achieve temperatures of a few millikelvins. The different formation processes of the two stable isotopes of helium produce the differing isotope abundances.īelow 0.8 K separate into two immiscible phases due to their dissimilarity (they follow different quantum statistics: 4Ītoms are fermions). Rocks from the Earth's crust have isotope ratios varying by as much as a factor of ten this is used in geology to investigate the origin of rocks and the composition of the Earth's mantle. 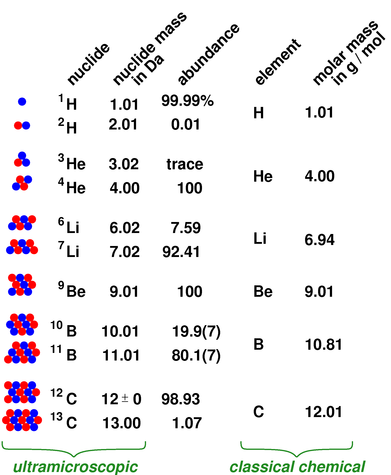
Is 1.62(29) ×10 −4, which is 121(22) times higher than that of atmospheric helium. In the Local Interstellar Cloud, the proportion of 3 However, the isotopic abundance of helium varies greatly depending on its origin. In the Earth's atmosphere, the ratio of 3 , with a half-life of 260(40) yoctoseconds ( 2.6(4) ×10 −22 s), although it is possible that 2 With a half-life of 806.92(24) milliseconds. All radioisotopes are short-lived, the longest-lived being 6 Although there are nine known isotopes of helium ( 2He) ( standard atomic weight: 4.002 602(2)), only helium-3 ( 3 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
